Whitepaper
Clinical Performance of the Spark UHPLC system
Overview
Liquid chromatography plays a central role in clinical laboratories, where analytical precision and reproducibility directly influence diagnostic reliability. In this study, an UHPLC system equipped with the Nexas autosampler, SPH1299 pump, and Sirocco column oven was evaluated under routine laboratory conditions using angiotensin converting enzyme (ACE) activity as a representative application.
Patient samples were analyzed, and the resulting chromatograms demonstrated highly stable retention times, baseline resolved separation of internal standard and analyte, and reproducible peaks even at low signal intensities. These findings indicate that the UHPLC system provides the instrumental and chromatographic stability required for enzymatic assays in clinical practice. Because ACE assays rely on kinetic enzymology and quantitative chromatography, both sensitive to system performance, they offer a meaningful model for evaluating UHPLC robustness in real-world laboratory conditions.
Mix & Dilute Functionality
The Mix & Dilute functionality of the Nexas Autosampler is designed to automate the preparation of diluted and mixed samples in a controlled and reproducible manner. The process consists of three fundamental steps: Add, Wait, and Mix, each contributing to accurate volume handling and proper homogenization of the sample. The sequence of these steps can be programmed in different orders, providing flexibility to adapt the workflow to specific application requirements. In addition, the functionality can be extended for Original Equipment Manufacturer (OEM) customers, providing targeted customization options engineered to integrate seamlessly with the exact technical requirements of their solutions. An example workflow, which is also used for generating the results presented in this technical note, is shown in Figure 1.
Angiotensin-Converting Enzyme (ACE) as a Model Analyte
Serum samples are incubated with the synthetic tripeptide substrate Hippuryl-Histidyl-Leucine (Hip-His-Leu). ACE cleaves the peptide bond, releasing hippuric acid and histidyl-leucine:
ACE + Hip-His-Leu → Hippuric Acid + His-Leu
After incubation, the reaction is stopped with hydrochloric acid, enabling kinetic determination of ACE activity. The amount of hippuric acid formed is directly proportional to the enzymatic activity present in the sample. Quantification is performed using UHPLC-UV detection at 228 nm. Benzoylalanine is added as an internal standard to correct for injection variability and matrix dependent signal fluctuations (2).
ACE activity determination is analytically demanding because it integrates biological sample handling, enzymatic kinetics, and quantitative chromatography. These characteristics make the assay sensitive to variations in injection precision, flow stability, and temperature control parameters directly linked to UHPLC system performance. As such, ACE activity provides a relevant and scientifically justified model for evaluating UHPLC robustness in routine laboratory conditions.
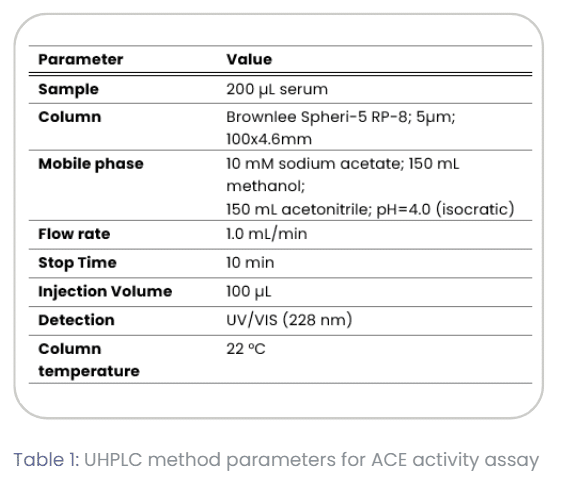
The UHPLC system was operated under conditions representative of daily clinical workflows at the University Medical Center Groningen (UMCG). Method parameters are summarized in Table 1.
Results
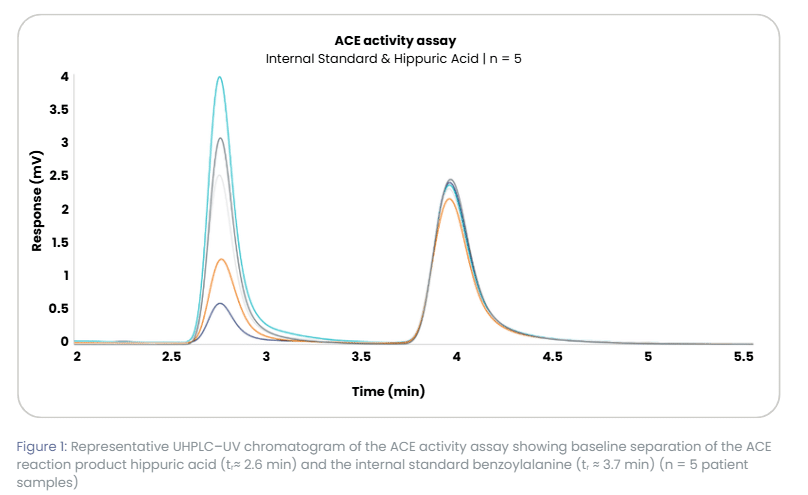
The overlay chromatogram of five patient samples shows two distinctly baseline separated peaks: hippuric acid as the ACE reaction product and benzoylalanine as the internal standard. Hippuric acid varies widely in peak height, reflecting true biological differences in ACE activity, while the internal standard remains highly consistent in retention time and peak shape. Only one sample shows a reduced internal standard response.
Such a decrease is expected and does not affect quantification. Because the internal standard is added at the start of sample preparation, any loss during extraction affects both analyte and internal standard proportionally. Results are therefore calculated from the analyte to internal standard ratio rather than absolute peak heights. In the sample with the lower internal standard peak, slightly more material was likely lost during work-up, but the ratio remains stable and the reported result is unaffected.
Overall, the chromatograms show tight overlay: retention times are reproducible, peak shapes are uniform, and internal standard performance is consistent. These features indicate stable pump flow rate, column temperature control, and detector response, demonstrating that the UHPLC system provides a robust analytical environment that captures true biological variability while maintaining the control needed for reliable quantification.
Conclusion
The UHPLC system equipped with the Nexas autosampler, SPH1299 pump, and Sirocco column oven demonstrated robust and reproducible performance in a clinically relevant ACE activity assay. The system delivered stable retention times, consistent peak shapes, and reliable quantification supported by internal standard normalization.
Because ACE activity determination integrates enzymatic kinetics with quantitative UHPLC-UV detection, it serves as a meaningful and sensitive model for evaluating UHPLC performance under routine laboratory conditions. The results of this evaluation support the system’s suitability for both clinical and non-clinical laboratories requiring dependable, high precision liquid chromatography.
Acknowledgements
Spark Holland thanks the staff of the University Medical Center Groningen (UMCG) for their collaboration and for contributing their time and expertise to this laboratory evaluation.
References
> Allen RK. A review of angiotensin converting enzyme in health and disease. Sarcoidosis. 1991 Sep;8(2):95-100. PMID: 1670005.
> Kwarts E, Beukenveld G, Gazendam J. Evaluation of a simple colorimetric assay for serum angiotensin-converting enzyme: comparison with a new ion-pair liquid chromatography-assisted assay. Ann Clin Biochem. 1982 Jul;19 (Pt 4):227-32. doi: 10.1177/000456328201900405. PMID: 6289722.
Click the button to download